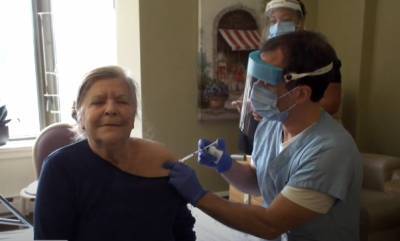
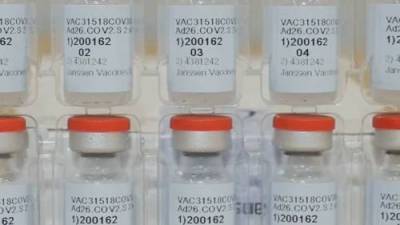
Vaccine makers to pledge 240 million doses by end of March
The U.S. could have 240 million doses of COVID-19 vaccines by the end of March. Pfizer and Moderna have pledged to have a combined 220 million doses available for shipment.
Johnson & Johnson, which has not yet been authorized by the U.S., has pledged to also have 20 million doses available by the end of March.
The company is slated to go before the Food and Drug Administration Friday for emergency use authorization. [TRENDING: HOA charges family $927 for trash on curb | Doctor faces hate crime charge in social distancing attack | Mandatory vaccines for first responders?] The vaccine makers, along with AstraZeneca and Novavax, are on Capitol Hill for hearings on Tuesday.