Pfizer begins testing COVID-19 booster shot with pneumococcal vaccine in older adults
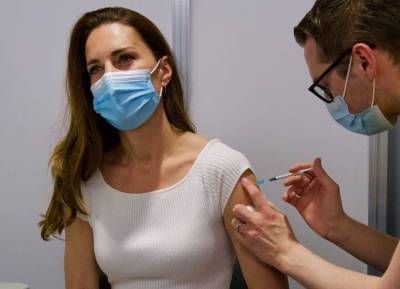
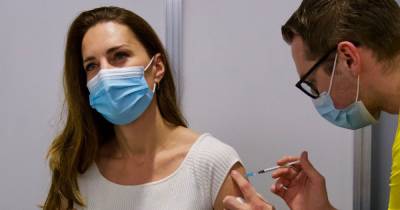
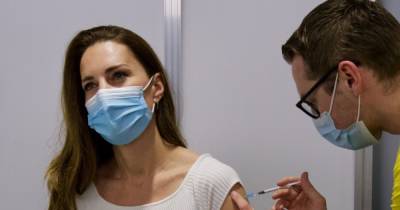
NEW YORK - Pfizer has started testing a booster shot of its COVID-19 vaccine in combination with a pneumococcal vaccine candidate, the company announced this week.
The company has enrolled 600 fully vaccinated adults age 65 or older for the trial, which aims to analyze the safety of when both vaccines are co-administered and the immune responses produced by each shot.The participants will have received their second dose of the Pfizer-BioNTech vaccine at least six months prior to entering the combination study.
Individuals in the study will receive either the company’s 20-valent pneumococcal conjugate vaccine candidate (20vPnC) plus a third dose of the Pfizer-BioNTech COVID-19 vaccine — referred to as a "booster" — or they’ll get the 20vPnC.