J&J’s single-dose Covid vaccine given approval for emergency use in India
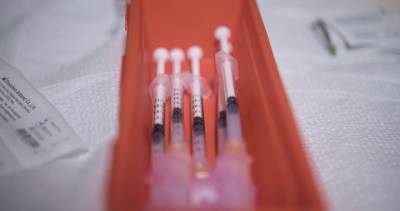
Johnson and Johnson’s single-dose COVID-19 vaccine is given approval for Emergency Use in India.Now India has 5 EUA vaccines.This will further boost our nation's collective fight against #COVID19 This will be the fifth Covid-19 vaccine to get approval after Covishield, Covaxin, Sputnik V and Moderna in India.
Earlier, a statement issued by the company on Thursday read, "On 5th August 2021 Johnson and Johnson Pvt. Ltd applied for Emergency Use Authorization (EUA) of its single-dose COVID-19 vaccine to the Government of India." The pharma company stated that it is an important milestone that paves the way to bringing the single-dose COVID-19 vaccine to the people of India, and the rest of the world, through a collaboration with Biological