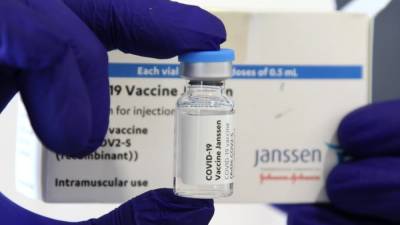
J&J applies with DCGI to conduct bridging study for Janssen covid jab in India
NEW DELHI : Johnson & Johnson said on Tuesday that it has applied with the Drugs Controller General of India (DCGI) V.G.
Somani for an approval to conduct a local bridging study of the company's single-dose Janssen covid-19 vaccine in the country.A spokesperson for the company said, the study was aimed to comply with local regulations and added that the application was in line with its commitment to provide safety and efficacy data necessary to support worldwide emergency use of the Janssen covid-19 vaccine candidate.However, the American company has as of now not applied seeking emergency authorization for import of its vaccine under revised regulations which expedited licensing to foreign covid-19 vaccine manufacturers that have an.