FDA adds J&J COVID-19 vaccine warning
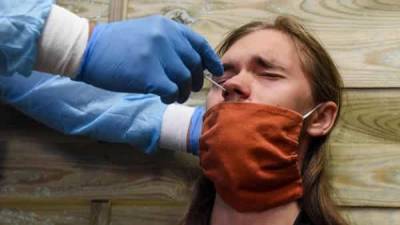
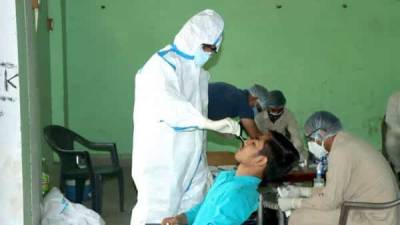
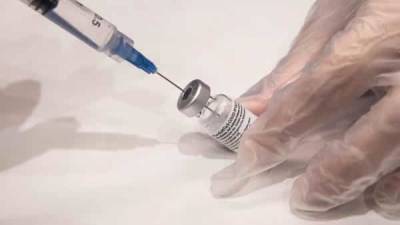
A Food and Drug Administration (FDA) fact sheet distributed with the Johnson & Johnson (J&J) vaccine will now contain warning language about a small increased risk that vaccine recipients have of developing Guillain-Barré syndrome (GBS), a neurologic disorder that can cause progressive, but usually reversible, paralysis.So far, the FDA has said there have been 100 cases identified in 12.8 million US J&J recipients, mostly in men over the age of 50.The FDA's fact sheet said patients who have developed GBS show symptoms within 42 days of inoculation, and said recipients should call health providers immediately if they develop numbness, difficulty walking, or loss of bladder control."Although the available evidence suggests an association