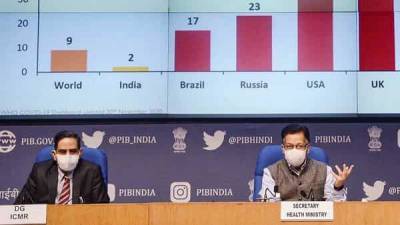
Serum event won’t affect covid vaccine timeline: govt
The serious adverse event reported during Serum Institute of India’s Covishield clinical trials will not delay the launch of India’s first coronavirus vaccine, the government said on Tuesday.The announcement comes amid concerns over the lack of transparency and insufficient disclosure about the neurological complications suffered by a 40-year-old trial participant from Chennai.“The adverse event will not affect the timelines in any manner whatsoever," said Union health secretary Rajesh Bhushan on Tuesday.The serious adverse event during the Covishield trial was the second such incident within a week.
Bharat Biotech International Ltd said it had seen an adverse event in August during clinical trials of its experimental vaccine,.