Serious adverse events rare after COVID-19 boosters in young kids
Data collected from two vaccine safety surveillance programs in the first 10 weeks of administration of third doses of the Pfizer/BioNTech COVID-19 to US children aged 5 to 11 years show that serious adverse events were rare.A related study in South Korea shows waning vaccine effectiveness (VE) in adolescents after two and three Pfizer doses but sufficient protection against critical illness.Most side effects mildIn the first study, published today in Morbidity and Mortality Weekly Report, researchers from the Centers for Disease Control and Prevention (CDC) analyzed adverse-event data from the agency's voluntary smart phone-based v-safe vaccine-monitoring program and the Vaccine Adverse Event Reporting System (VAERS) from May 17 to Jul 31, 2022.
VAERS is a passive vaccine surveillance system managed by the CDC and the Food and Drug Administration (FDA).On May 17, the FDA authorized Pfizer COVID-19 boosters for children aged 5 to 11 who had received two doses of the same vaccine at least 5 months earlier.During the study period, v-safe received information about 3,249 third doses of the Pfizer vaccine in this age-group.
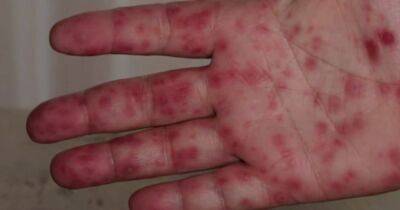
The most common adverse reactions included injection-site pain (66.7%), fatigue (28.9%), and headache (19.9%). Most reactions were reported the day after vaccine receipt and were mild.After a third dose, injection-site and systemic reactions were often reported (68.5% and 45.6%, respectively).
Injection-site and systemic reactions had been documented in similar proportions after the second dose (68.0% and 45.8%, respectively).While most adverse effects reported by those who experienced pain, fatigue, headache, or muscle pain were mild, more moderate and severe symptoms were reported after the third dose