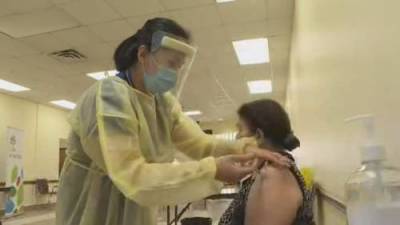
Pfizer COVID-19 vaccine authorized for children 5-11, U.S. FDA says
COVID-19 vaccine in children aged five to 11 years old, setting the stage for a mass vaccination campaign for young children across the United States.The Pfizer pediatric vaccine is just one-third of the strength of the dosage given to adults and teens.
It will be given as a two-dose series, with the doses three weeks apart.The FDA’s authorization comes shortly after its expert panel unanimously recommended approval of the vaccine for this age group, saying the potential benefits outweighed any risks of side effects, including a rare heart complication known as myocarditis that has been reported in some teens and young adults who received a stronger dose.