fox29.com
93%
902
Paxlovid: Biden administration expands availability of COVID antiviral pill
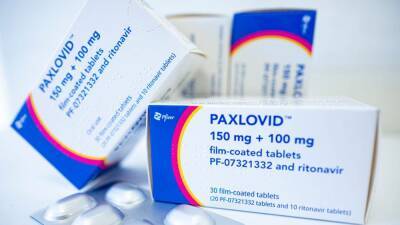
Photo of Paxlovid, a life-saving COVID-19 antiviral treatment, manufactured by Pfizer (Photo by Fabian Sommer/picture alliance via Getty Images) WASHINGTON - President Joe Biden's administration is taking steps to expand availability of the life-saving COVID-19 antiviral treatment Paxlovid, as it seeks to reassure doctors that there is ample supply for people at high risk of severe illness or death from the virus.Paxlovid, produced by Pfizer, was first approved in December. Supply of the regimen was initially very limited, but as COVID-19 cases across the country have fallen and manufacturing has increased it is now far more abundant.