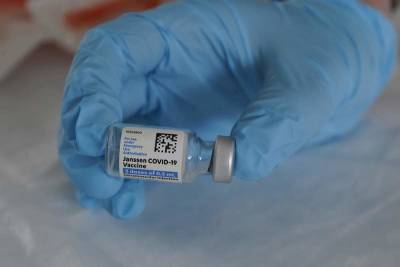
J&J COVID-19 vaccine pause: What to know if you received shot, have future appointment and more
WASHINGTON - On Tuesday, U.S. officials recommended a "pause" on the use of the Johnson & Johnson COVID-19 vaccine in order to investigate U.S.
reports of a rare type of blood clot following vaccination.Nearly 7 million J&J doses have been administered in the U.S., almost all without serious side effects.
Now, the millions of people who have received the shot - and those who have been scheduled to - are left with many questions surrounding the safety of the vaccine.
Here are some answers to common questions about the Johnson & Johnson COVID-19 vaccine and the recommended "pause" by the U.S.