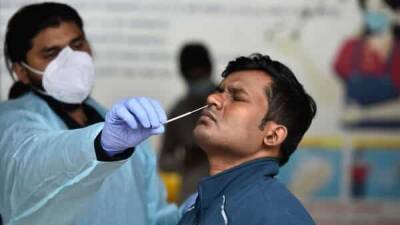
India's first coronavirus vaccine: Human trials of Covaxin on 375 people starting soon
Covaxin, India's first possible vaccine against COVID-19, will be tested on over 1,100 people in two phases, according to a report in Bloomberg.
Bharat Biotech, an unlisted Indian vaccine maker, received regulatory approval to start human clinical trials for its experimental shot.
The phase 1 trial of Covaxine is scheduled to start next week. The company has planned to enroll 375 people in the first phase of clinical trials, an Indian Council for Medical Research (ICMR) spokesperson told Bloomberg.
The company has set July 13 as the last date of enrollment for first trial.The ICMR has selected 12 institutes to conduct these trials, including AIIMS in Delhi and Patna.