FDA OKs just 10M J&J vaccine doses from troubled Baltimore plant
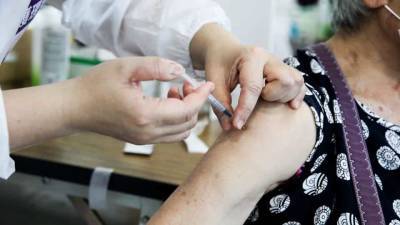
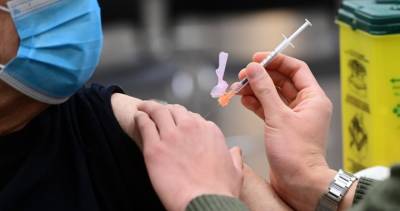
BALTIMORE - The U.S. Food and Drug Administration announced Friday it will authorize the release of two batches of Johnson & Johnson’s COVID-19 vaccine manufactured at its troubled Baltimore plant.According to the Associated Press, the authorized batches will include as many as 10 million doses of vaccine.
But they said many more must be thrown out.Baltimore’s Emergent BioSolutions factory, known as Bayview, is one of several contractors for J&J that produces its one-shot vaccine in bulk.
The concentrated vaccine then is shipped to other factories for final steps, including diluting them to the correct strength, putting them in vials and packaging them up, the AP reported.RELATED: FDA finds multiple ‘quality issues’ at Johnson & Johnson.