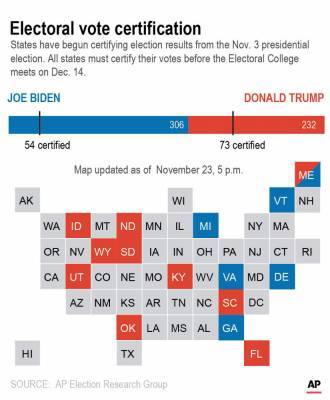
FDA approves emergency use of Regeneron COVID-19 monoclonal antibody therapy used to treat President Trump
FILE - View of Corporate and Research and Development Headquarters of Regeneron Pharmaceuticals, Inc. (Lev Radin/Pacific Press/LightRocket via Getty Images)The U.S.
Food and Drug Administration has issued an emergency use authorization (EUA) of two more monoclonal antibody treatments for mild cases of COVID-19, according to a news release.
The same dual therapy was used to treat President Donald Trump when he was diagnosed with COVID-19 in October, the FDA confirmed.Monoclonal antibodies are laboratory-made proteins that mimic the immune system’s ability to fight off harmful pathogens such as the novel coronavirus, according to the FDA.