FDA acts to expand use of treatment for COVID-19 patients with mild-to-moderate disease
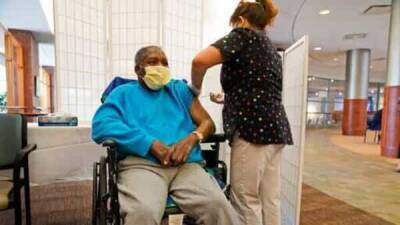
to certain non-hospitalized adults and pediatric patients for the treatment of mild-to-moderate COVID-19 disease.Previously, the use of the Gilead Sciences Inc.
drug was limited to those requiring hospitalization."On the heels of the FDA’s recent authorization of two oral antiviral drugs, today’s actions bolster the arsenal of therapeutics to treat COVID-19 and respond to the surge of the omicron variant," Patrizia Cavazzoni, director of the FDA’s Center for Drug Evaluation and Research, said in a news release. "Today’s actions provide adults and pediatric patients, with mild-to-moderate COVID-19 who are at high risk of severe COVID-19, with a treatment option they could receive outside of a traditional inpatient hospital setting, including at skilled nursing facilities, home healthcare settings and outpatient facilities such as infusion centers," she added.The agency has expanded the approved indication for Veklury to include its use in adults and pediatric patients 12 years and older – and weighing at least 88 pounds – with positive results of direct SARS-CoV-2 viral testing and who are not hospitalized, have mild-to-moderate COVID-19 and are at high risk for progression to severe disease.RELATED: Gilead says it expects remdesivir to remain effective against omicron variantThe FDA also revised Veklury's emergency use authorization (EUA) to also authorize the drug for treatment of pediatric patients less than 12 years of age, weighing at least 3.5 kilograms to less than 88 pounds, with the same conditions.High-risk, non-hospitalized patients may receive Veklury via intravenous infusion for a period of three days.The FDA said that the approval of Veklury for use in these patients is supported by a randomized,.