European regulators say AstraZeneca vaccine safe, but can't rule out rare events
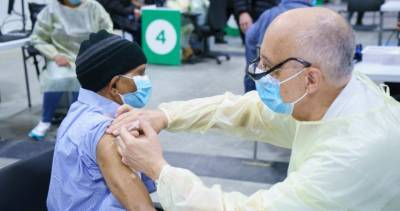
The European Medicines Agency (EMA) safety committee today released the conclusions of its preliminary probe of blood clotting reports in people who received the AstraZeneca-Oxford COVID-19 vaccine.
The committee said immunization benefits still outweigh the risks, but may be linked to extremely rare cases of clots linked to low blood platelets, especially in those younger than 55.The eagerly awaited findings come in the wake of vaccine program suspensions in nearly 20 countries, mostly in Europe.
Countries such as Austria, Norway, and Germany reported a small number of suspected side effects, prompting the pauses, ordered mainly as a precaution during the EMA's investigation.