EU reviews J&J vaccine for rare nerve disorder after US warning
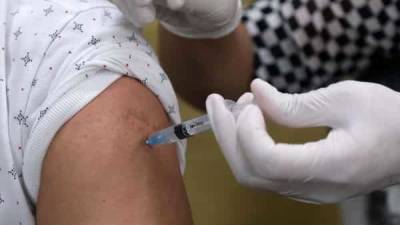
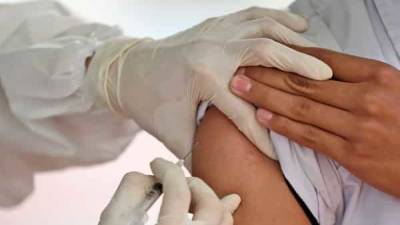
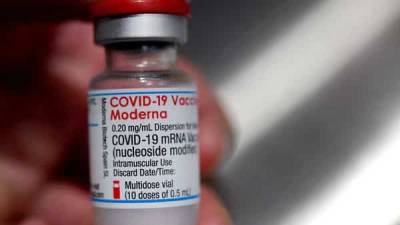
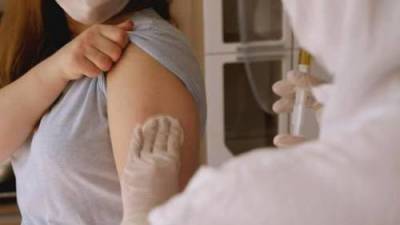
The European Medicines Agency (EMA) said it was analysing data on rare cases of a nerve disorder reported among recipients of Janssen (Johnson & Johnson's) Covid-19 vaccine, after the United States added a warning label to the shot.
The "EMA's safety committee (PRAC) is analysing data provided by (J&J) ... on cases of Guillain-Barre syndrome (GBS) reported following vaccination," the European medicine regulator said in a statement to Reuters. "PRAC has requested (Johnson & Johnson) to provide further detailed data." The EMA scrutiny comes after the US Food and Drug Administration (FDA) yesterday added a warning for GBS to the fact sheet for J&J's shot, and after the EMA four days ago included a warning for AstraZeneca's Covid-19 shot for