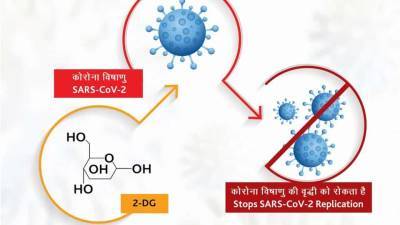
EMA begins assessing Pfizer-BioNTech jab for 12-15 year olds
The European Medicines Agency has said it has begun evaluating the use of Pfizer-BioNTech's Covid-19 vaccine for those aged 12 to 15, after a similar request in the United States.
Its possible authorisation for children is seen as a crucial step towards achieving herd immunity. The EMA's human medicines committee "will carry out an accelerated assessment of data submitted by the company that markets Comirnaty," said an EMA statement - referring to vaccine's brand name.
This included results from a large ongoing clinical study involving adolescents from 12 years of age, the Amsterdam-based agency said.
The agency would announce its findings in June "unless supplementary information is needed", the statement added. Pfizer and BioNTech on