CDC signs off on Johnson & Johnson's single-dose COVID-19 vaccine
Centers for Disease Control and Prevention's Advisory Committee on Immunization Practices (ACIP) has voted to recommend Johnson & Johnson's COVID-19 vaccine for emergency use for individuals ages 18 and older in the United States.The 12-0 vote, with one recusal for a potential conflict of interest, comes following an official emergency use authorization by the Food and Drug Administration on Saturday.
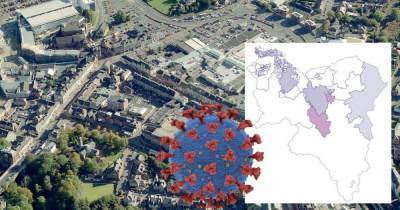
The ACIP, a group of independent health experts that advises the CDC on the types of populations and circumstances for which vaccines should be used, made the recommendation after an emergency session was called to review the vaccine's data.RELATED: Johnson & Johnson's single-shot COVID-19 vaccine endorsed by FDA expert panelCDC Director Dr..