Beware if you are taking this COVID vaccine! You suffer from severe allergic reactions
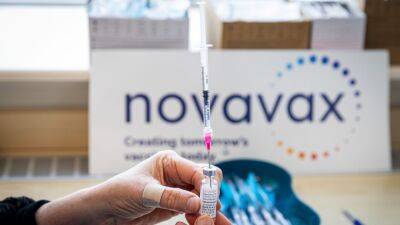
Pfizer and Moderna vaccines. For now, the FDA authorised Novavax's initial two-dose series for people 18 and older. “I encourage anyone who is eligible for, but has not yet received, a COVID-19 vaccine to consider doing so," FDA Commissioner Dr.
Robert Califf said in a statement. Before shots begin, the Centres for Disease Control and Prevention must recommend how they should be used, a decision expected next week.
Only 250,000 doses of Novavax's COVID vaccine, Nuvaxovid, have been administered in Europe so far since its launch in December, according to the European Centre for Disease Prevention and Control.
The Novavax vaccine is made of copies of the spike protein that coats the coronavirus, packaged into nanoparticles that to the immune system resemble a virus.