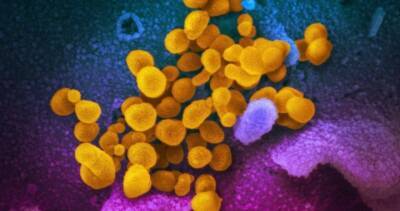
AstraZeneca says late-stage trials show coronavirus vaccine ‘highly effective’
[ Sign up for our Health IQ newsletter for the latest coronavirus updates ]All three vaccines must be approved by regulators before they can be widely distributed.The results come as a second wave of COVID-19 hits many countries, once again shutting businesses, restricting social interaction and pummeling the world economy.AstraZeneca said it will immediately apply for early approval of the vaccine where possible, and it will seek an emergency use listing from the World Health Organization, so it can make the vaccine available in low-income countries.The AstraZeneca trial looked at two different dosing regimens.